PLEASE CLICK HERE TO LOGIN CMDRD
China Medical Device Regulatory Database (CMDRD) is an easy-to-use web-based regulatory database on medical device in China, it enables user to determine the regulatory obligations under CFDA regulations for their medical device (IVD included) and get the solutions to comply with it via using the CMDRD’s sub-database:
- MDCSS-Medical Devices Classification Search System
- MDCS-Medical Devices Classification System
- MDRGD-Medical Device Regulations and Guidance Database
- MDSSS-Medical Devices Standards Search System
- MDRSC-Medical Devices Regulatory Supporting Center
Benefits from CMDRD
- Classify your medical device
- Define the technical requirement for your product
- Determine the regulatory obligations
- Tracking CFDA issues
- Get the practical advice on regulatory compliance
- Supported by professional RA specialist online
- Removing language and cultural barrier
How it works
| Requests | CMDRD | Solutions |
| Determine the classification for a medical device | Classification Database | MDCSS | Searching the classification database by using MDCSS |
| Classification Rule | MDCS | Classify your product by yourself via using MDCS | |
| Define the technical requirement for a medical device | Product Standards Database | MDSSS | Searching the standard database by using MDSSS |
| Determine the regulatory obligations in China | Regulations and Guideline Database | MDRGD | Tracking medical device regulations and guideline by using MDRGD |
| Get the regulatory support online | Supported online | MDRSC | Supported by medical device regulatory expertise services in MDRSC |
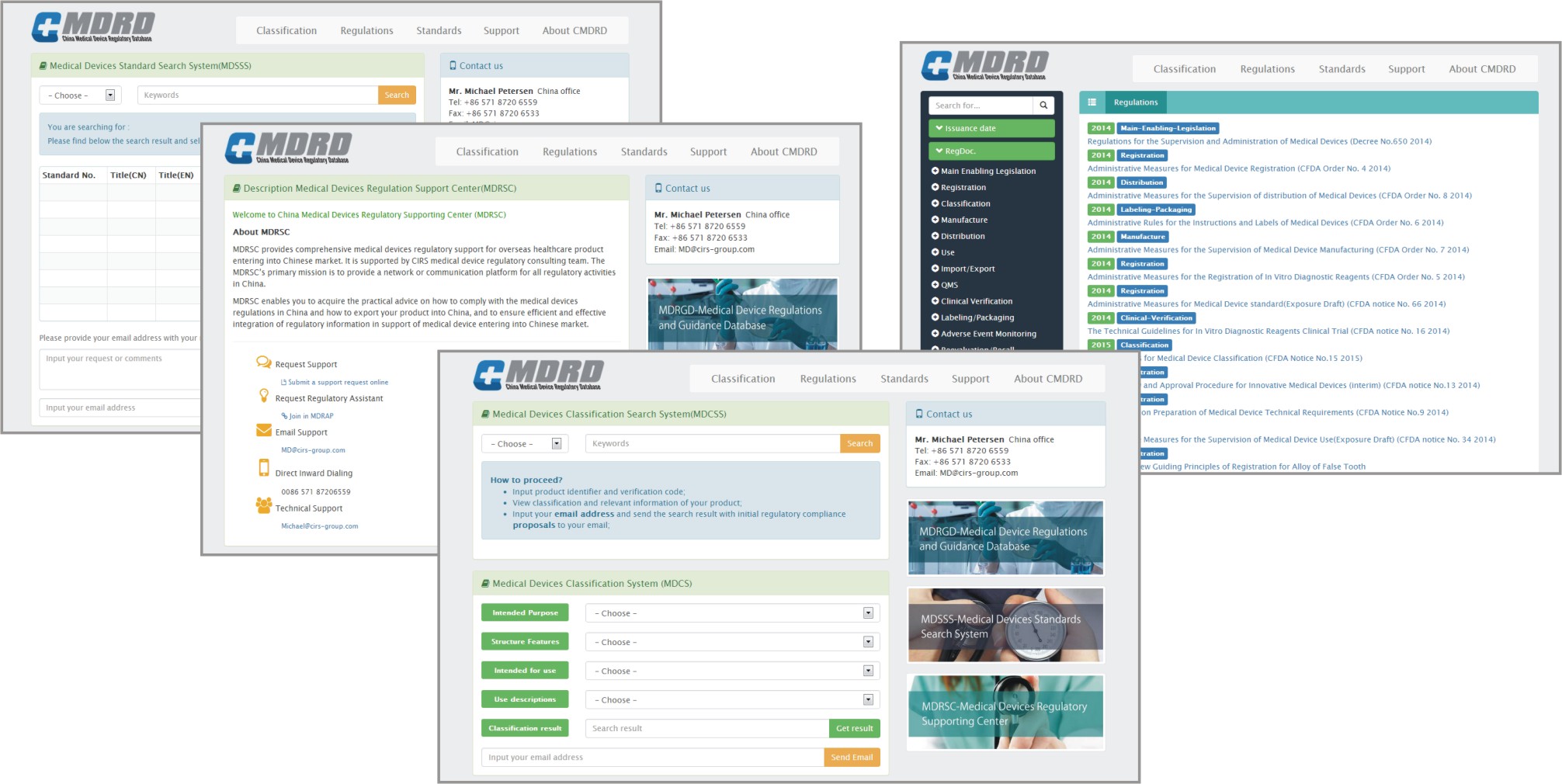
CMDRD provides a simple, easy-to-use interface for quick access to medical device classification, regulation, standard and other important information.
Medical Devices Classification Search System (MDCSS)
The MDCSS contains over 4000 types of medical devices which approved in Chinese market. There are included class I, class II, class III medical devices as well as product which not considered as medical devices (Non medical devices).
Class I Medical Devices are those with low risk degree and for which safety and effectiveness can be ensured through routine administration
Class II Medical Devices are those with moderate risk and for which strict administration is required to ensure their safety and effectiveness
Class III Medical Devices are those with higher risk and for which must be strictly controlled via special measures in respect to safety and effectiveness
Non Medical Devices are those not managed as medical device under CFDA related regulations, or it doesn’t meet the definition of medical devices.
Medical Devices are defined as any instrument, apparatus, appliance, material, in vitro diagnostic reagents and calibration substances and other similar substances and related articles, including the needed computer software. Its main effectiveness is achieved via physics ways and so on. It does not achieve its principal action in or on the human body by means of pharmacology, immunology or metabolism, but which may be assisted in its function by such means; the use of which is to achieve the following intended objectives:
1. Diagnosis, prevention, monitoring, treatment or alleviation of disease;
2. Diagnosis, monitoring, treatment, alleviation of or compensation for an injury or handicap conditions;
3. Investigation, replacement or modification for anatomy or a physiological process;
4. Life support or maintenance;
5.Control of conception;
6. Offer information for medical or diagnosis purpose via inspecting the human samples.
What does it do?
MDCSS lists the product name, classification name, classification codes, intended purpose, structure features, and the managing category. The medical device name and classification codes identify the generic category of medical device for CFDA and the classification assigned to a medical device is based upon the classification rule of medical device designated under the Regulations for the Supervision and Administration of Medical Device (State Council Decree No. 650).
MDCSS enables you to determine the managing category/class of medical device with regulatory controls by classification code, classification name or product name. MDCSS also provides initial assessment of your regulatory obligations under CFDA regulations. CIRS is constantly maintaining its database to keep it up to date.
How it works?
- Input product identifier and verification code;
- View classification and relevant information of your product;
- Input your email address and send the search result with initial regulatory compliance proposals to your email;
What does it do?
The classification should be determined by the combined judgment from four aspects: intended purpose, structural features, intended for use and use descriptions. MDCS enables you to determine the managing category for your product by screening the factors of intended purpose, structural features, intended for use and use descriptions. MDCS also provides initial assessment of your regulatory obligations under CFDA regulations. CIRS is constantly maintaining its rules to keep it up to date.
How it works?
- Select the intended purpose of your product, and
- Select the structural feature of your product, and
- Select the intended for use of your product, and
- Select the use descriptions of your product, then
- View classification (managing category ) of your product
- Input your email address and send the classification result with initial registration proposals to your email;
- Main Enabling Legislation
- Registration
- Classification
- Manufacture
- Distribution
- Use
- Import/Export
- QMS
- Clinical Verification
- Labeling/Packaging
- Adverse Event Monitoring
- Reevaluation/Recall
- Administrative Supervision
- Medical Device Inventory
What does it do?
The MDRGD contains the text of medical device regulations, guidance and related technical measures, as well as the implementation information and the practical know how to comply with it. The MDRGD enables you to determine the following conditions:
- What product are applicable to China medical device supervision procedures
- What is the legal obligations under CFDA regulations
- The special regulations for overseas applicant
- The data requirements and application dossiers for overseas applicant should be prepared
- The implementation timeline and the deadline
- The background of the regulations (revision history)
- Who are the regulatory authorities for medical device approval of overseas product
- Practical know how to comply with CFDA regulations
- How to prepare the technical files for medical device registration in China.
- Expert suggestions for overseas applicant to acquire the relevant approval from CFDA
- Screening the relevant regulations by keywords, RegDoc. or issuance date
- Select the target regulations and click into,
- View the relevant information: summary, history, timeline, documents and follow up
- Join in the China Medical Device Regulatory Assistant Program(MDRAP) for practical advices
What does it do?
The MDSSS enables you to search the relevant standards and technical guideline by standards number and name, and apply for the text from CIRS Medical Devices Regulatory Supporting Center (MDRSC).
How it works?
- Input standard number or name;
- View the standard information (number, name, IDT, effective date etc.,);
- Input your email address and send the search request to CIRS experts;
- CIRS expert will keep response to you up your request.
MDRSC enables you to acquire the practical advice on how to comply with the medical devices regulations in China and how to export your product into China, and to ensure efficient and effective integration of regulatory information in support of medical device entering into Chinese market.
back to top







